Author(s): Wohlgemuth, Walter A.
Author(s): Wohlgemuth, Walter A.
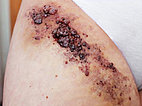
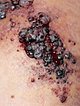
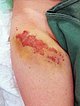
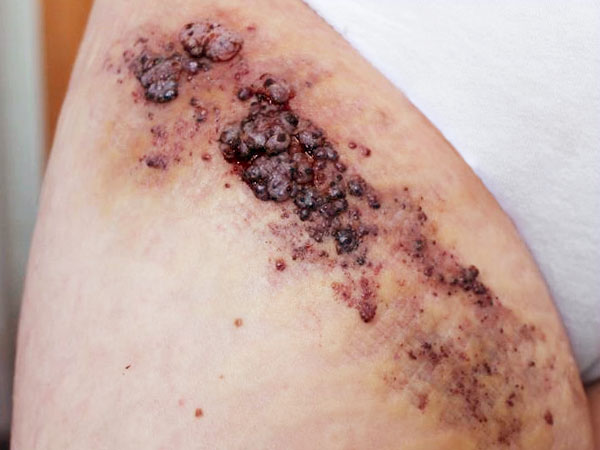
Photograph of a 15-year-old girl with recurrent swelling and pain ventrally in the right groin and proximal thigh. In this painful area, crust-like or multiple clustered, dark purple-colored vesicles repeatedly appear on the skin. From these vesicles, blood-tinged lymph fluid is discharged (lymphorrhea), on some days up to 500 ml per day. Clinically, this is a classic picture of a cutaneous lymphatic malformation with lymphangioma circumscriptum due to microcystic parts of the lymphatic malformation extending into the skin.
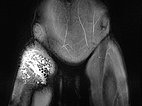
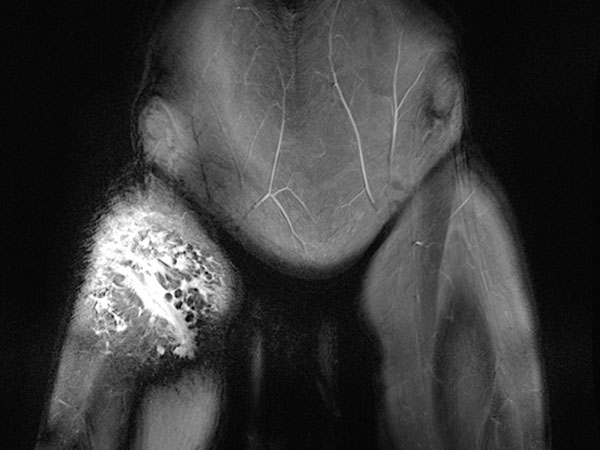
On coronal T2-weighted, fat-suppressed MRI at the level of the proximal thigh, the lymphatic malformation is highly hyperintense with a net-like distribution because of the interstitial fluid (lymph) it contains.
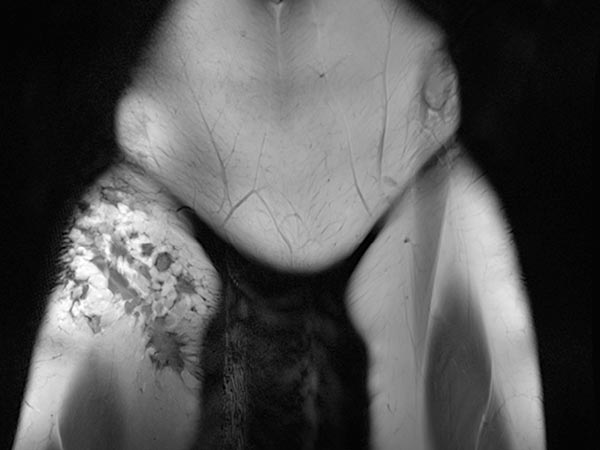
The same MRI plane in non-enhanced T1-weighted image shows the lymphatic malformation as hypointense in hyperintense subcutaneous adipose tissue, corresponding to contained lymphatic fluid.
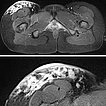
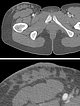
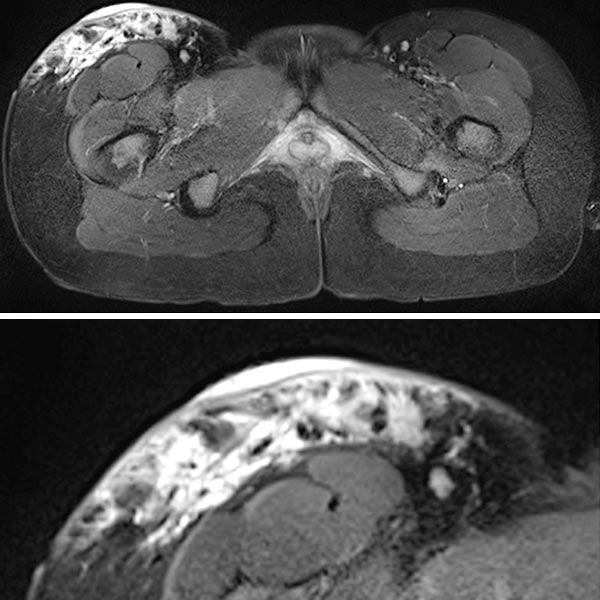
In transverse T2-weighted, fat suppressed MRI, the purely epifascial extension of the lymphatic malformation in the subcutaneous adipose tissue is clearly demonstrated. The underlying musculature is not affected.
Note the exophytic extension of the microcystic portions (highly hyperintense) of the lymphatic malformation in the detail enlargement (bottom image) extending to above the level of the skin.
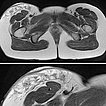
In the same MRI plane (top overview, bottom detail enlargement) in T2-weighted imaging without fat suppression, the coexisting connective tissue parts of the hard palpable lymphatic malformation are better seen as a hypointense reticular pattern. Note again the raised exophytic portion above the skin level.
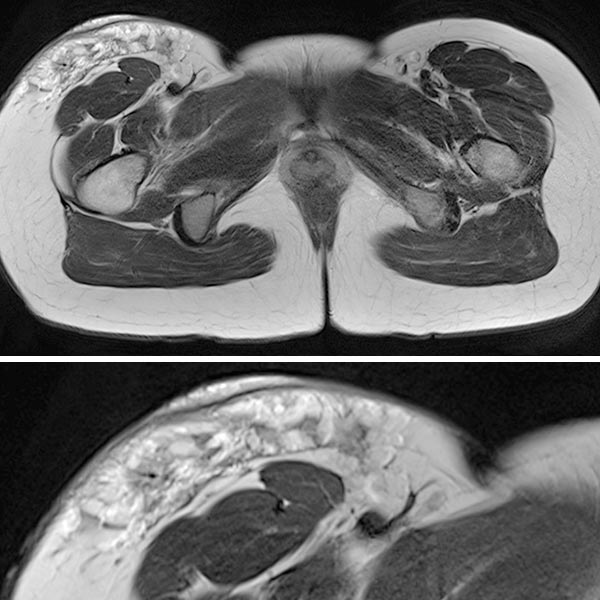
In the axial fat-suppressed, T1-weighted images after contrast administration (top overview, bottom detail), the strongest enhancement is found in the cutaneously located, exophytic, microcystic portions in the skin. In contrast, the underlying lymphatic malformation in the subcutaneous adipose tissue hardly enhances.
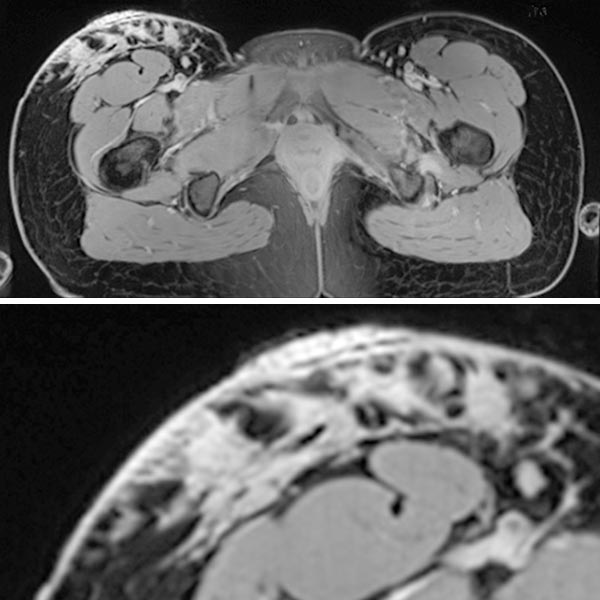
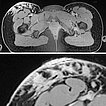
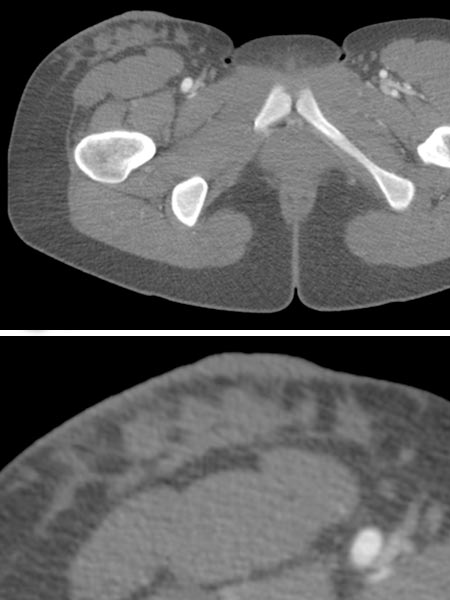
In the corresponding computed tomography, the lymphatic malformation with its fluid deposition and connective tissue component appears somewhat more hyperdense than the surrounding subcutaneous adipose tissue. The overall soft tissue contrast is poorer than in MRI. There is also no contrast enhancement on CT. Note in the detail enlargement (bottom image) the thickening of the skin due to the cutaneous microcystic portions of the LM.
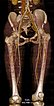
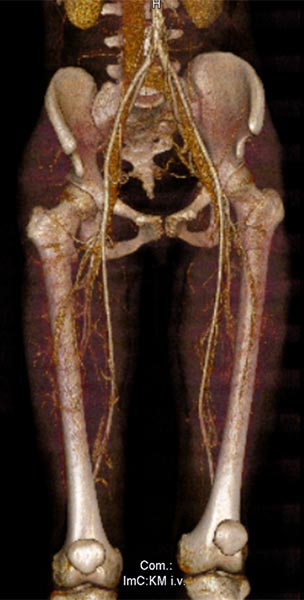
In the 3D reconstruction of the computed tomography angiogram, there are normal findings for the right-sided arteries and veins of the groin and thigh. These are not involved in the malformation. The underlying bony structures are also unremarkable.
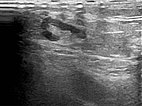
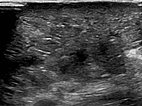
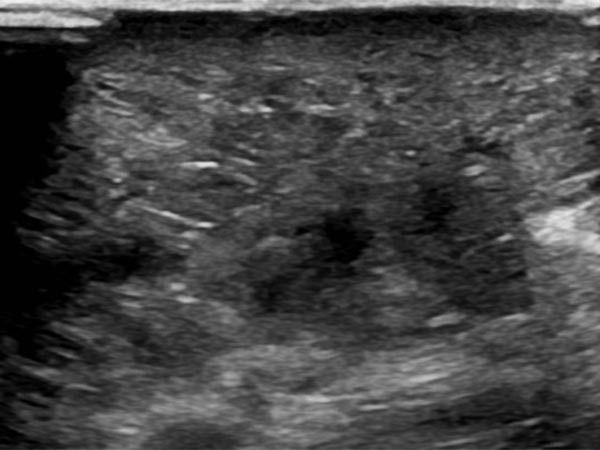
B-scan ultrasonography of the subcutaneous portion of the lymphatic malformation shows a dysplastic dilated lymphatic duct as an anechoic tubular structure. The surrounding subcutaneous adipose tissue is clearly more echogenic than normal due to the interspersed lymphatic fluid.
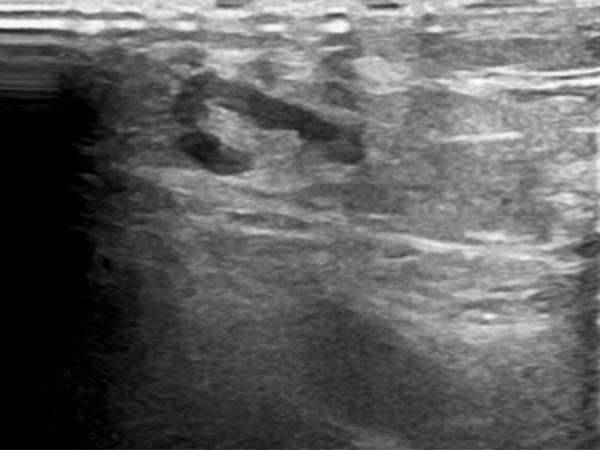
B-scan ultrasonography (cross-section of proximal ventral thigh) also shows the clearly echogenic tissue, interspersed with individual very fine, dilated, fluid-filled lymphatic ducts.
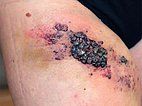
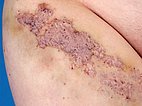
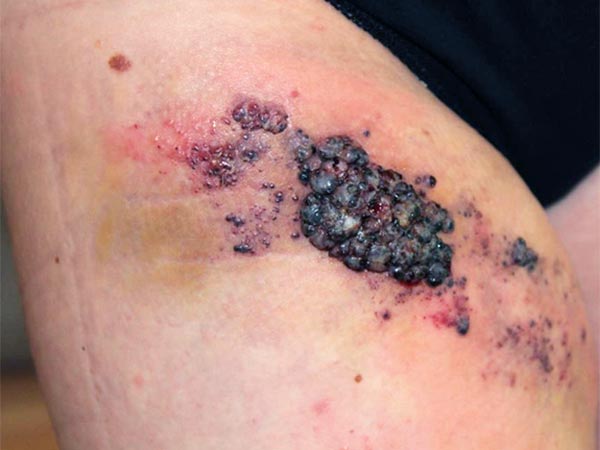
As a result of the permanent lymphorrhea, the natural barrier function of the skin against bacteria is lacking. Streptococcal infections in the form of erysipelas occur again and again. This is visible here as increasing local swelling and especially redness of the skin. The redness is also very painful, and the patient currently has a fever.
Episodes of blood-tinged lymphorrhea (skin discharge of a mixture of blood and lymph fluid) also continue to recur. The established therapeutic plan is to be escalated in steps. First, an attempt should be made to seal the skin by sclerotherapy using picibanil (OK-432), for example, in a direct puncture technique so that the lymphorrhea is reduced and the erysipelas become less frequent.
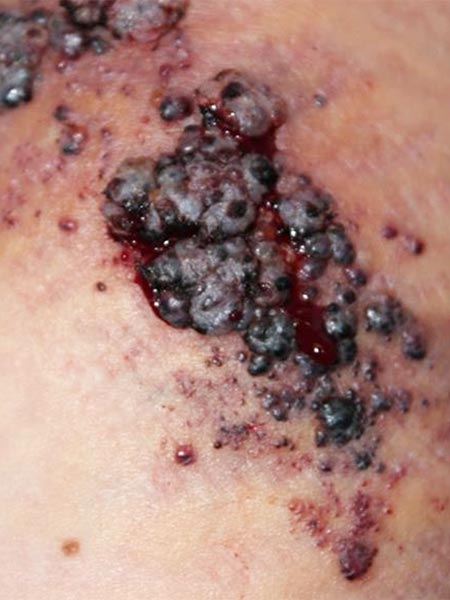
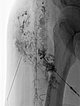
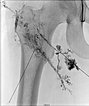
Image of direct lymphography with water-soluble contrast medium in direct puncture technique under sonographic guidance.
Here, it was possible to puncture the dysplastic, dilated lymphatic channels of the lymphatic malformation in the subcutaneous tissue several times.
Picibanil (OK-432) was then injected into these channels as a sclerosing agent to seal the cutaneous dysplastic lymphatic channels via the inflammation.
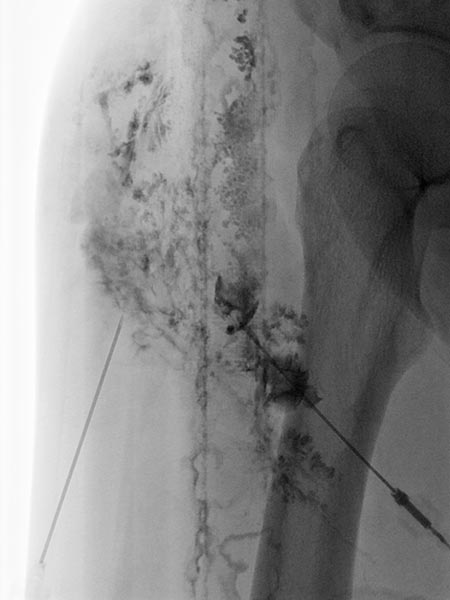
Lymphography in direct puncture technique under sonographic guidance.
The subcutaneous lymphatic vessel system is severely dysplastic, partially enlarged and showing bizarre net-like dilation. Normal lymphatic vessels are significantly smaller.
Again, picibanil (OK-432) was injected for inflammation-induced lymphatic vessel sclerotherapy and skin sealing.
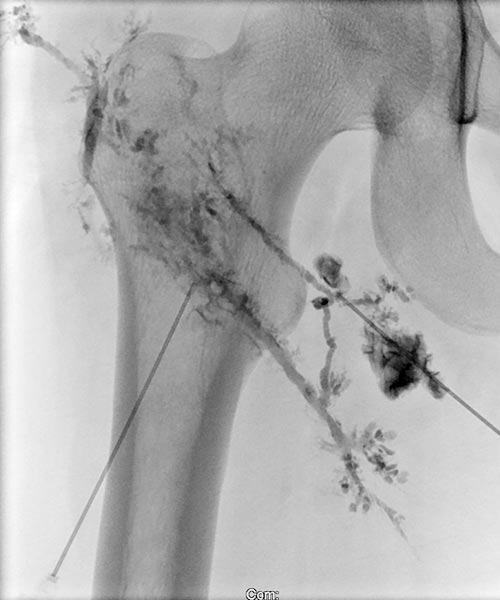
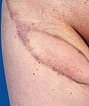
In a further step, the remaining parts of the lymphangioma circumscriptum on the skin were ablated by CO2 laser. The sealing of the skin was initially very successful thanks to the combination of sclerotherapy and laser treatment.
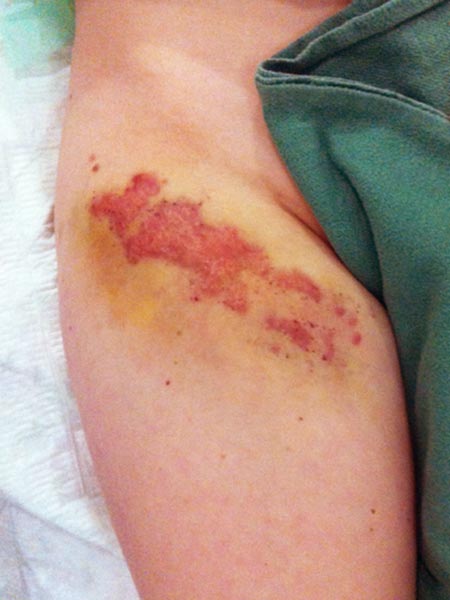
The intermediate functional result after this combination therapy was initially satisfactory. However, the underlying portions of the lymphatic malformation lead to a risk of recurrence in the long term.
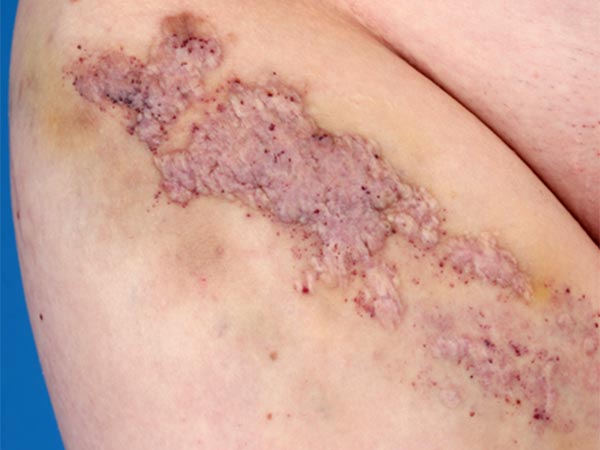
Therefore, the next step was open surgical resection as far as possible, plastic-surgical coverage with a microvascular graft and creation of a lymphovenous anastomosis.
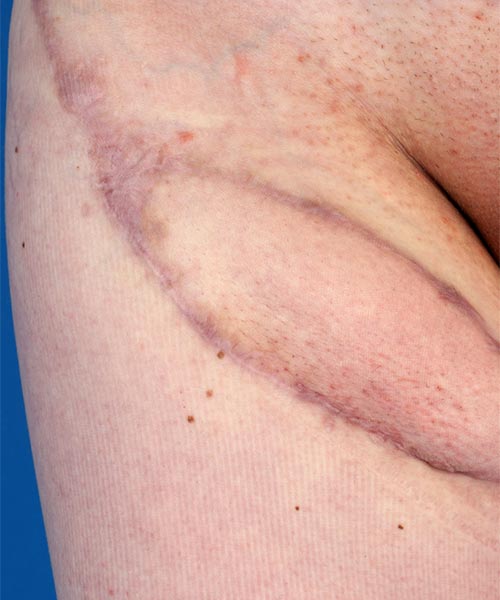
Skin involvement of a lymphatic malformation (LM), in this case in the form of cutaneous microcystic parts appearing exophytically in multiple clusters of small lymphatic vesicles (lymphangioma circumscriptum), is particularly difficult to treat in the long term and prone to recurrence. Direct resection is usually not possible because of the risk of permanent lymphatic leakage, and recurrences may also occur on the transferred pedicled microvascular graft. A permanent cure can often be achieved even in these cases by a combination of different therapeutic procedures in stages (in this case sclerotherapy, laser therapy and open resection).
Published: 2020
All images © Wohlgemuth