Author(s): Wohlgemuth, Walter A. | Dießel, Linda | Kisser, Ulrich
Author(s): Wohlgemuth, Walter A. | Dießel, Linda | Kisser, Ulrich
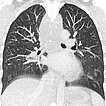
33-year-old female patient, since childhood recurrent attacks of "pseudocroup" with pronounced respiratory stridor. Previously known "hemangioma" of unclear origin in the neck. In an old thoracic CT, extensive mass in the neck, hypopharynx, larynx, down to the mediastinum. Multiple phleboliths in the mediastinum are clearly visible, indicating a venous malformation.
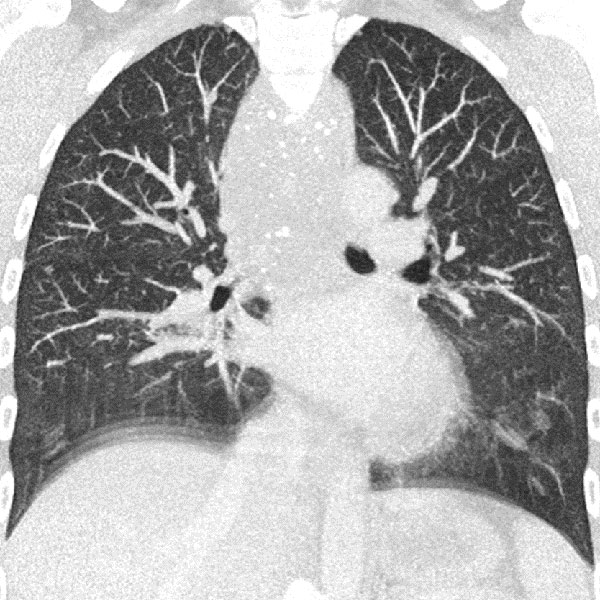
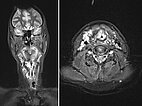
In connection with a systemic infection, now occlusion of the upper airway due to an inflammation-related increase in the swelling of the venous malformation; intubation became necessary. A biopsy was performed. On MRI in the coronal plane (T2-weighted, fat-suppressed), the lesion is very clearly visible as a strongly hyperintense mass. Extensive lesions in the hypopharynx and larynx on both sides.
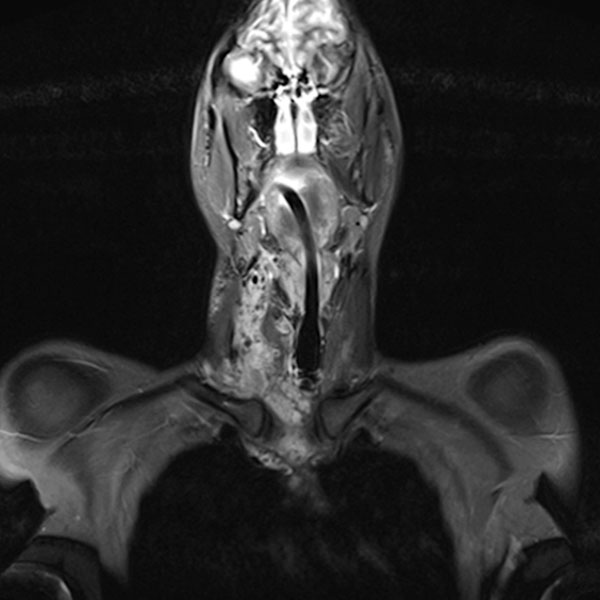
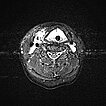
Transverse T2-weighted, fat-suppressed MRI of the neck also shows extensive, grossly hyperintense (white) portions of the venous malformation on the larynx and neck. These obstruct the upper airway. The endotracheal tube is visible in the center.
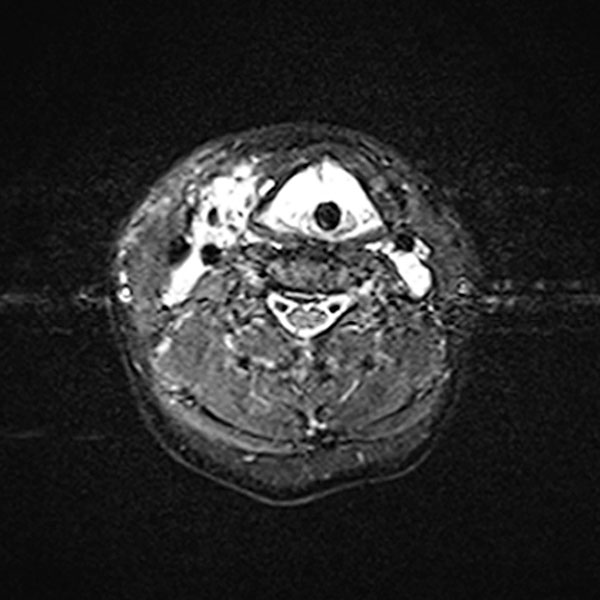
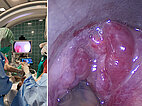
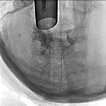
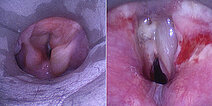
Thus it was decided to sclerose the portions of the venous malformation directly obstructing the airway in the area of the larynx. In biplanar angiography, the rigid laryngoscope is first used to visualize the situation (left image). In the endolarynx, the vocal cords are completely obscured by supraglottic parts of the venous malformation and are no longer visible.
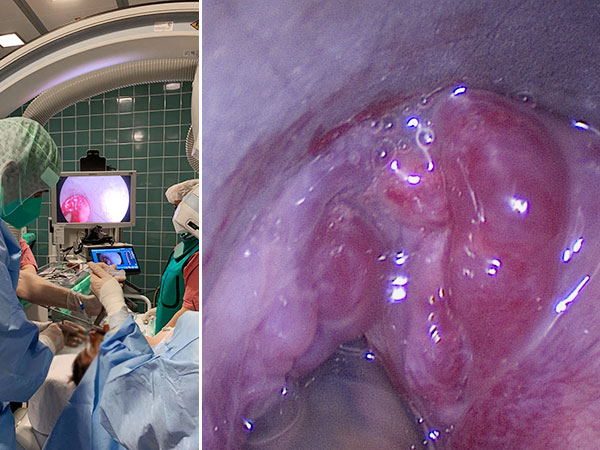
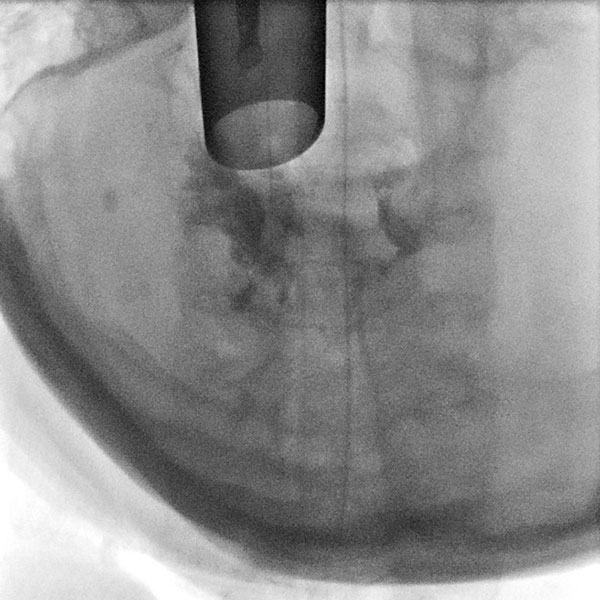
For sclerotherapy, a butterfly needle is advanced through the rigid laryngoscope using grasping forceps into the venous malformation for direct puncture. After aspiration of blood, contrast medium is injected to prove needle placement in the venous malformation before injection of the sclerosing agent.
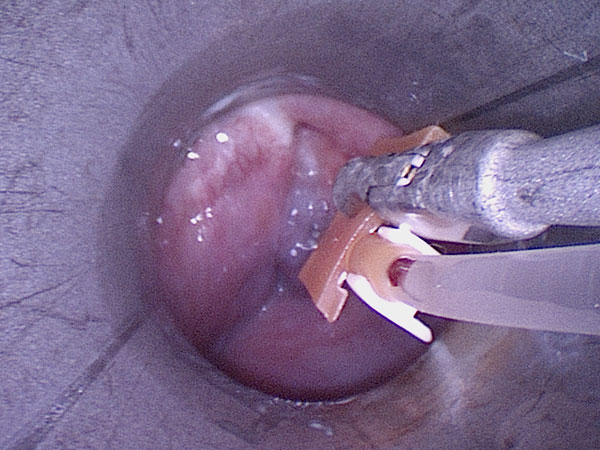
Control fluoroscopy after contrast injection shows correct needle placement in the venous malformation without outflow via a major communicating vein. The venous malformation can thus be well sclerosed via this position.
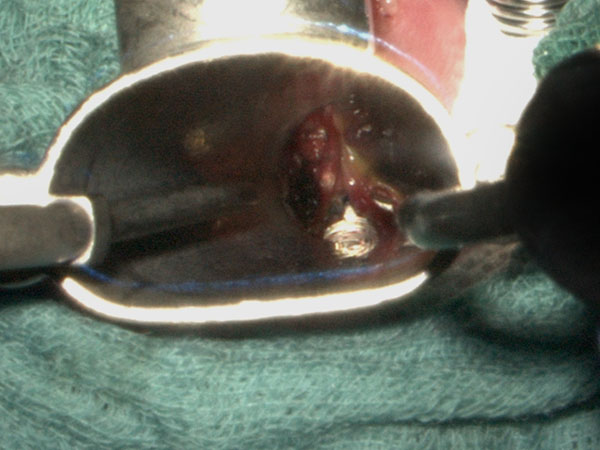
To improve the local situation directly at the level of the vocal cords, CO2 laser vaporization of parts of the venous malformation directly attached to the vocal cords is performed using a Kleinsasser endoscope.
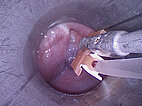
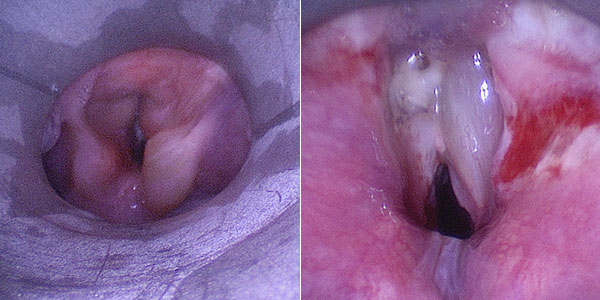
The control laryngoscopy 10 days later shows a clear improvement of the local situation in the pharynx and larynx, especially directly at the level of the vocal cords. These are visible again and not displaced by the space-occupying venous malformation.
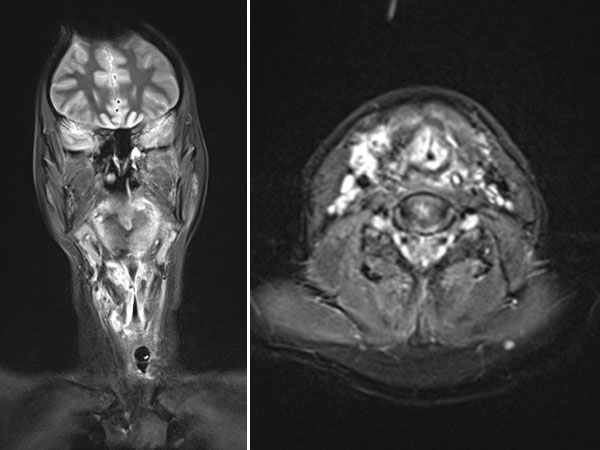
MRI now also shows a marked improvement. The space-occupying effect due to the venous malformation (in this image white = strongly hyperintense on coronal and transverse, T2-weighted, fat-suppressed MRI) has diminished. The upper airway, although still narrow, is clear for spontaneous breathing.
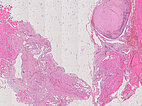
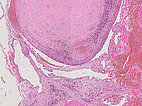
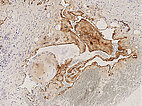
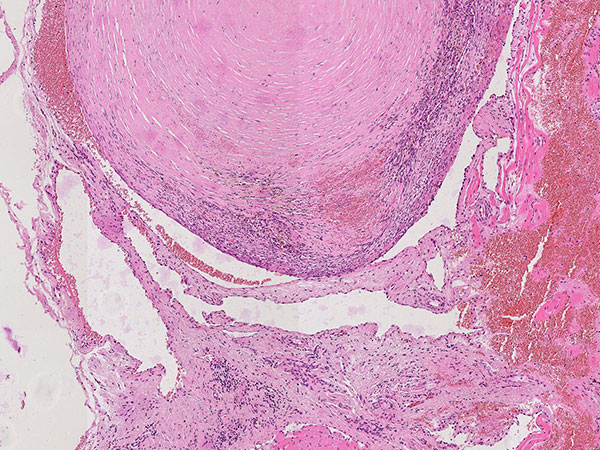
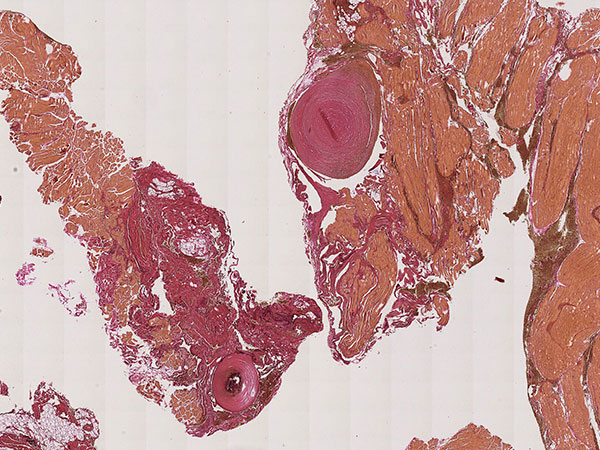
Hematoxylin & eosin stain, 21× magnification: A phlebolith already transformed into connective tissue is shown top right. Overall, as an indication of a vascular lesion, increased irregular vascular cavities are visible in the histopathological overview image.
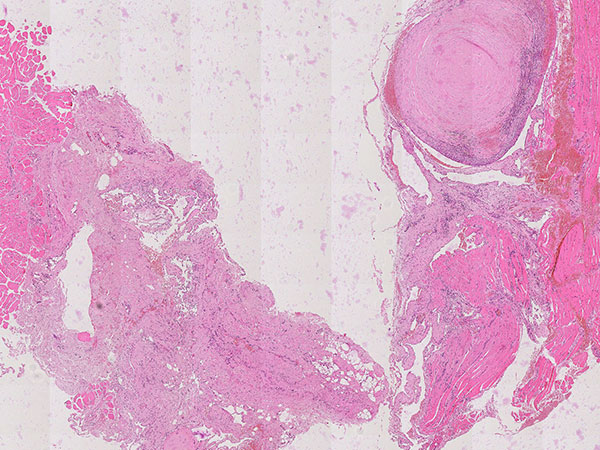
Hematoxylin & eosin stain, 64x magnification: Layered, predominantly connective tissue appearance of the older, partially organized phlebolith. The phlebolith is surrounded by multiple proliferated, irregularly structured, thin-walled vascular spaces.
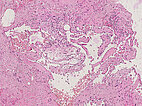
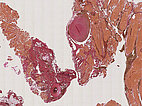
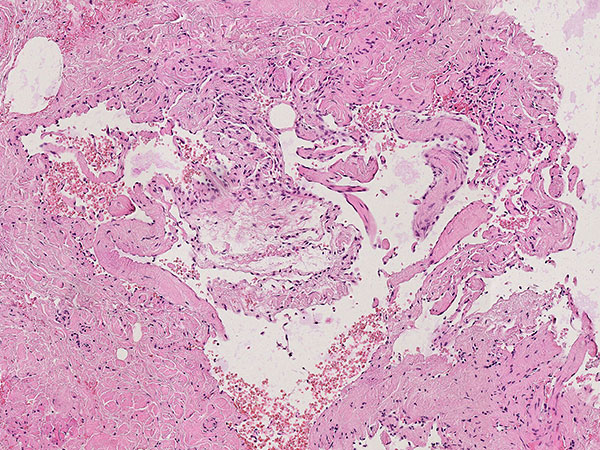
Hematoxylin & eosin stain, 100× magnification, histopathological section: Within the irregular vascular cavities, which are clearly increased in the lesion, some erythrocytes are still visible that were not washed out during preparation. In contrast to lymphatic malformations which contain lymphatic fluid, a venous malformation is filled with blood (visible as erythrocytes). Thus, a venous malformation is more likely here than a similar-looking lymphatic malformation.
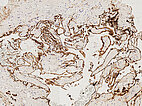
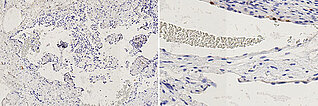
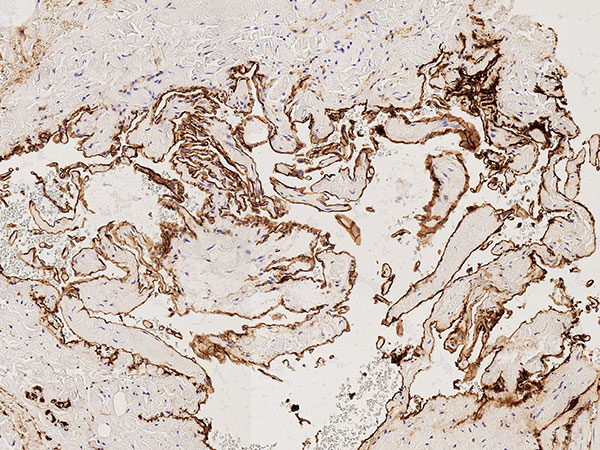
Histopathology, CD31 staining, 110× magnification: Strong staining (dark brown-stained cells) of the entire border of the irregular, vascular dysplastic cavities with the vascular endothelium-specific marker CD31 clearly indicates a vascular malformation. Only a very thin vessel wall is detectable beneath the vascular endothelium.
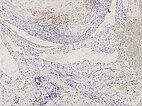
Histopathology, D2-40 stain, 95× magnification: The vascular endothelial lining of the multiple irregular vascular spaces does not stain with podoplanin (D2-40). Thus, they are not lymphatic vascular endothelia. In conjunction with the wall structure and the shape of the lesion, this is another clear indication of a venous malformation (not a lymphatic malformation).
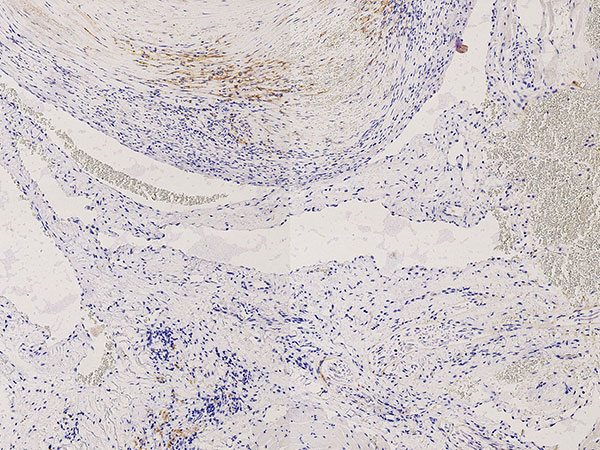
Histopathology, SMA stain ("smooth muscle actin"), 100x magnification: In this stain, the smooth musculature (smooth muscle actin; here smooth muscle cells of the vessel wall) is stained dark brown. This shows the highly irregular, asymmetric and in some cases even absent wall structure of the venous malformation. This is clearly dysplastic compared to a regular normal vein wall.
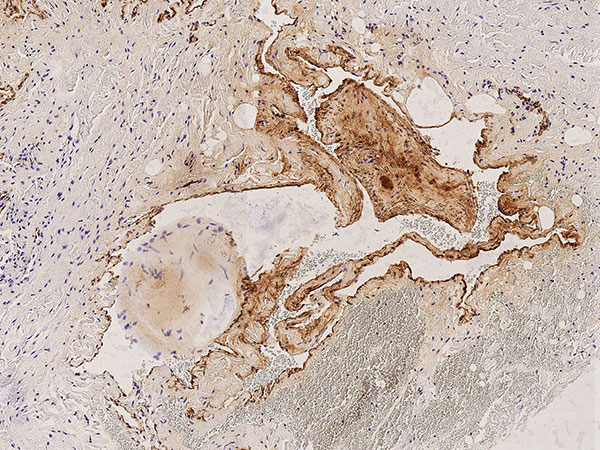
Histopathology, EvG stain, 20x magnification: The Elastica van Gieson connective tissue stain again illustrates the dysplastic wall structure of the venous malformation with yellow-stained smooth muscle fiber tracts and surrounding skeletal muscle intermixed with red-stained connective tissue areas and interspersed delicate black elastic fibers (barely visible in the overview). This appearance is typical of venous vessels. The irregular structure shown here is atypical and indicates the malformed vein wall structure. Two phleboliths are remodeled into connective tissue.
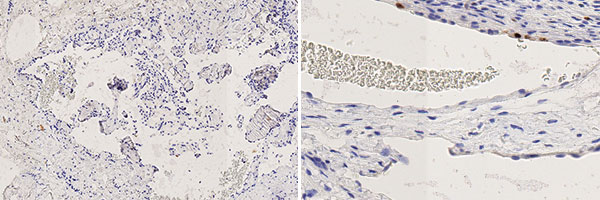
Histopathology, Ki67 staining, magnification 130x on the left and 260x on the right. As a proliferation marker, Ki67 indicates all cells undergoing division in the broadest sense (outside the G0 phase in the cell cycle). In the present venous malformation, only very few actively dividing nuclei (here without mitotic spindles) are detectable as positive nuclear staining. The very low proliferation rate is thus an indication of a benign lesion.
The swelling on the patient's neck, which had been known since childhood, was initially interpreted as a "hemangioma". In fact, histopathologically and from the clinical and imaging point of view, it is a venous malformation. During the course of an infection, more pronounced thrombophlebitis has obviously occurred within the venous malformation with increased swelling. The upper airways, which were already very narrow at the level of the larynx due to the malformation, were thus obstructed, making intubation necessary.
The histopathological workup of a tissue biopsy of the patient showed the classic histopathological findings of a venous malformation. It is not a lymphatic malformation because the lesion is filled with erythrocytes and not with lymph, but especially because of negativity of the vascular endothelia to D2-40 (podoplanin). This was obvious because the lesion also contained multiple phleboliths, which are virtually pathognomonic for venous malformations.
Finally, therapy management was interdisciplinary, involving ENT, radiology, anesthesiology and pathology.
In a targeted approach the stenosing parts were sclerosed and parts on the vocal cords were vaporized directly with CO2 laser.
Equivalent obstructions of the upper airway due to venous or lymphatic malformation are not uncommon and require a combined endoscopic approach in addition to close interdisciplinary coordination.
Published: 2022
All images © Wohlgemuth/Dießel/Kisser