Author(s): Wohlgemuth, Walter A. | Dießel, Linda
Author(s): Wohlgemuth, Walter A. | Dießel, Linda
Photograph of the lesion on the labium majus, right more than left, at the age of 3 months. At that time still interpreted as a possible infantile hemangioma. Multiple accompanying teleangiectasias, the lesion is rather dark-colored for an infantile hemangioma.
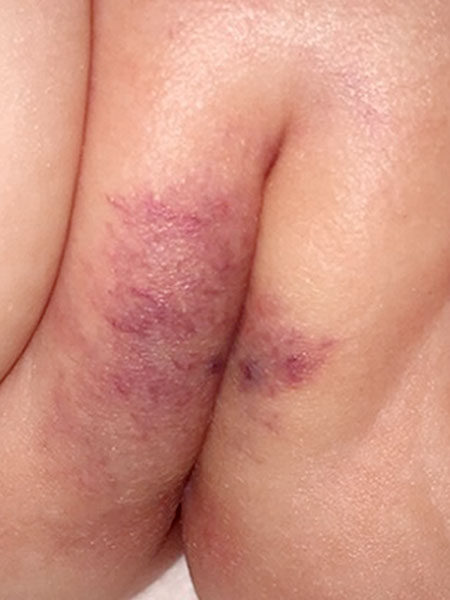
Photograph of the lesion on the labium majus, right more than left, at the age of 14 months. Raised, circumscribed, soft blue swellings of about 5 mm have developed centrally on both sides. The picture certainly does not correspond to an infantile hemangioma anymore. The color of the lesion is darker, more like purple.
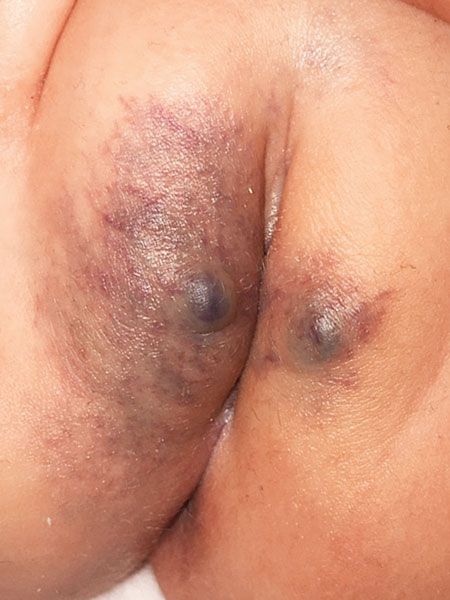
In the 22nd month of life, the coloring of the lesion is rather blue (initially more reddish). The two circumscribed, here cone-like raised parts have become progressively larger.
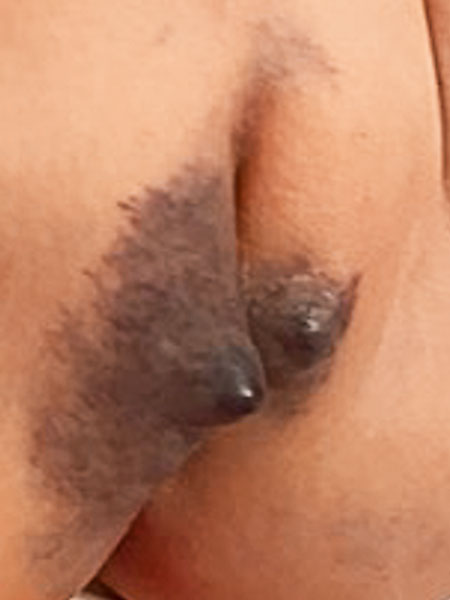
Subsequently (32nd month of life) there has been further massive progression. In particular, the entire right labium majus was now visible as a blue, sponge-like swelling, asymmetrically enlarged. Since the differential diagnosis from another (vascular) tumor could not be established with certainty at this time, an MRI was performed.
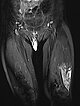
In coronal, T2-weighted, fat-suppressed MRI at 32 months of age, the characteristic strongly hyperintense (= white) signal of a slow-flow vascular malformation is found in the labial lesion. In addition, a similarly strong hyperintense signal in an intramuscular lesion is seen in the vastus lateralis muscle of the left thigh. Thus, a vascular malformation is very likely.
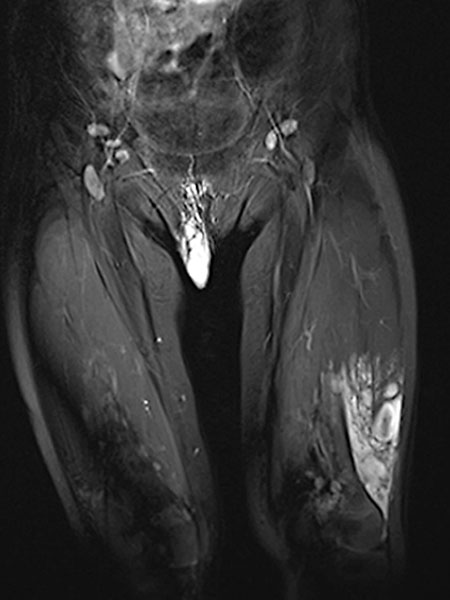
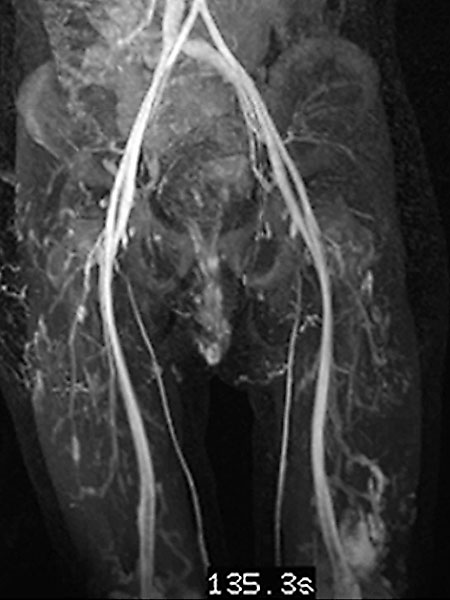
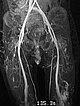
In the late phase of dynamic, high temporal resolution, contrast-enhanced MR angiography 135 seconds after i.v. contrast administration, there is slow, delayed contrast pooling in the lesion. Thus, an arteriovenous malformation with its rapid flow can be clearly excluded. Additional slow contrast pooling of the second lesion on the lateral distal left thigh.
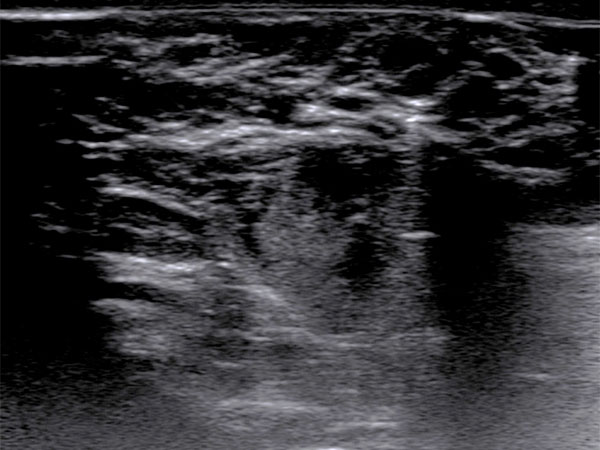
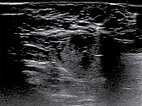
To confirm the diagnosis, an ultrasound-guided punch biopsy was now taken from the lesion on the right labium majus using a 16-gauge disposable biopsy gun. The biopsy needle advanced into the lesion produces strong distal acoustic shadowing on B-scan imaging.
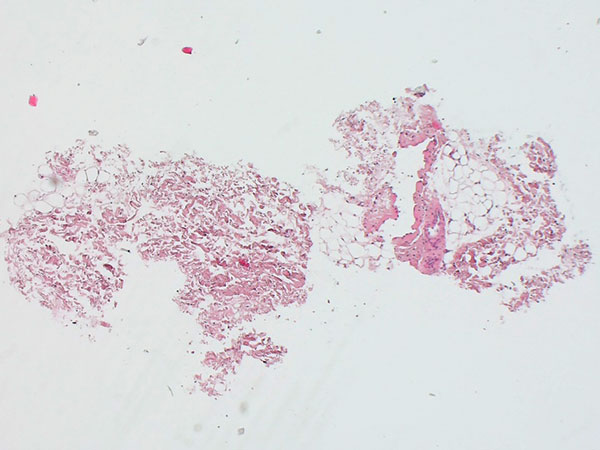
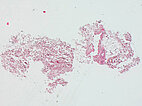
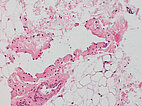
Histopathological image, hematoxylin & eosin stain (HE), here 25-fold overview magnification: A dysplastic, vein-like irregular vascular cavity is found. This is embedded in normal adipose tissue and stroma.
Histopathological image, hematoxylin & eosin stain (HE), here in 200x magnification: An "empty space" lined with endothelium with single, centrally contained erythrocytes and a malformed, thin venous vessel wall is visible. The "empty space" is bordered on the outside by vascular endothelial cells. A normal arteriole is visible next to it.
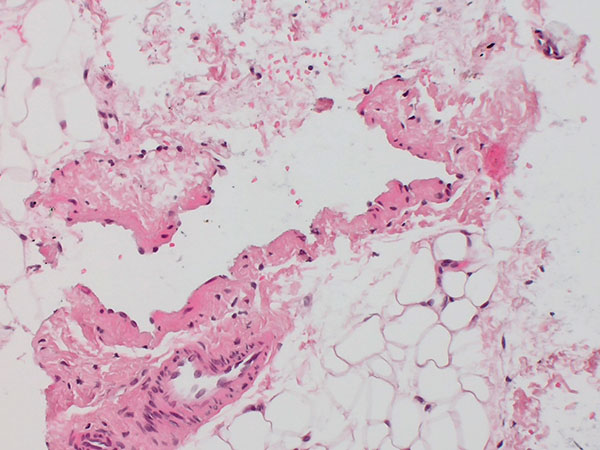
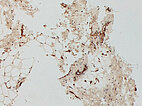
Histopathological image, CD31 stain, 100× magnification: With this vascular endothelium-specific immunohistochemical CD31 stain, vascular endothelial cells are stained black-brown. The vascular endothelia (brown), which are clearly increased in number in the lesion, correspond to the irregular venous vascular spaces of the venous malformation. In addition, nonspecific small vessels.
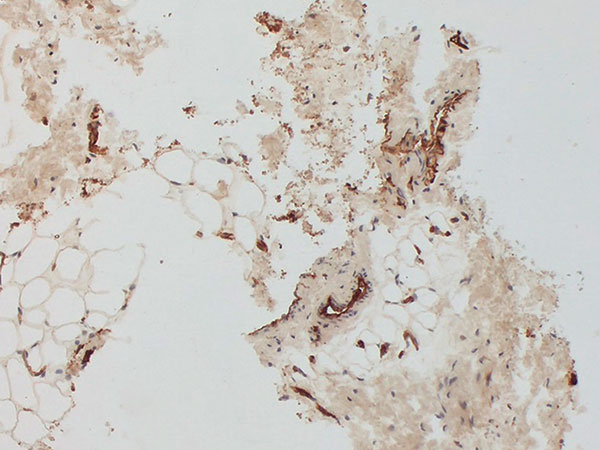
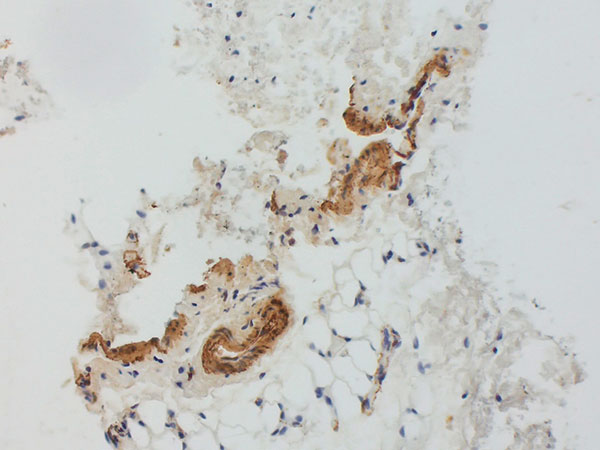
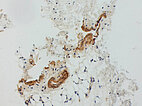
Histopathological image, SMA stain, 100x magnification: The SMA ("smooth muscle actin") stain does not show the normal, ordered smooth muscle wall structure of venous vessels. Rather, the lesion is surrounded by asymmetric and irregular SMA-positive (brown-stained cells) smooth muscle as a result of the malformation in vessel maturation. This is quite typical of venous malformations. The diagnosis was therefore confirmed histopathologically.
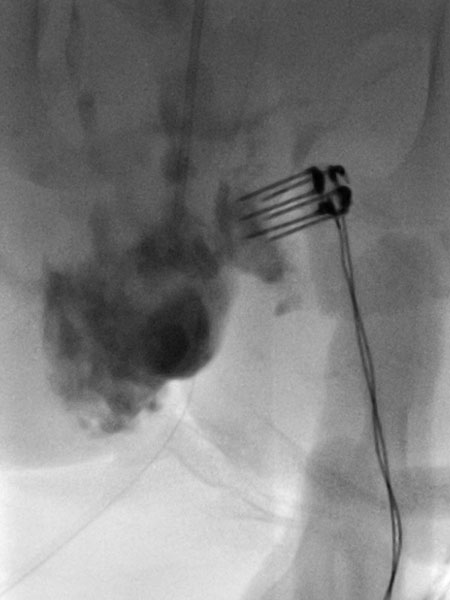
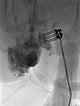
After confirmation of the diagnosis, bleomycin electrosclerotherapy was performed. For this purpose, the venous malformation was first punctured on both sides with a needle under ultrasound guidance and injected with contrast medium. Good needle placement, no major drainage veins were found.
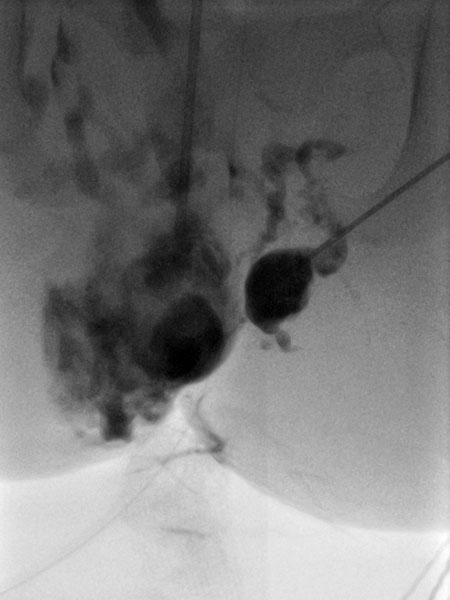
A total of 0.75 mg bleomycin (diluted one to three in contrast medium) was now injected directly into the venous malformation via the two horizontal needles. Subsequently, a total of 11 applications of reversible electroporation with finger electrode with 15 mm active needle length were performed on both sides.
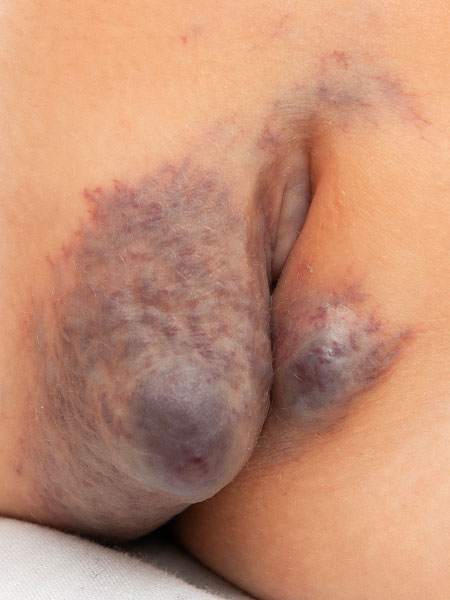
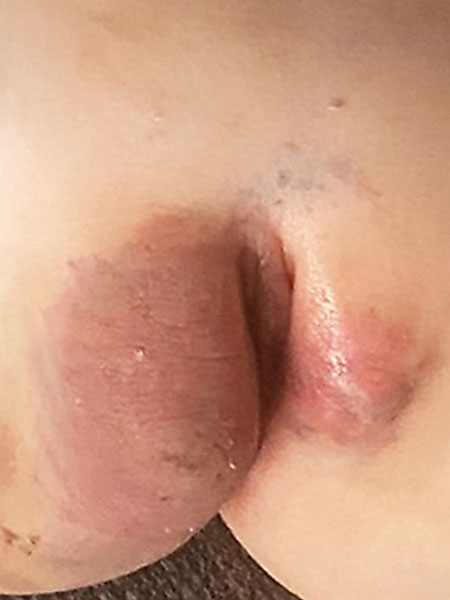
The image 5 days after bleomycin electrosclerotherapy (BEST) still shows significant swelling, especially of the right labium majus, and moderate hematoma formation from the puncture. This is consistent with a normal postoperative course. On account of the swelling and reduced blood filling, the color of the lesion is now slightly redder again rather than the typical blue of the venous malformation.
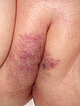
Over the next few weeks, the swelling quickly receded completely. In the follow-up examination approximately 2 months postoperatively after bleomycin electrosclerotherapy, the overall findings are clearly improved. The swelling has completely resolved. In addition, the two cone-like, blue exophytic parts of the venous malformations are no longer visible. Overall, the skin coloration caused by the venous malformation has also improved and become paler.
![Venous malformation on the labium majus [Translate to English:] GenitaleVenous malformation on the labium majus venöse Malformation](/fileadmin/images/patientenbeispiele/43-vm-labium-majus/f43-15-venoese-malformation.jpg)
The patient's history is rather less typical, given that venous malformations do not usually show such early and rapid progression. In view of this striking course, an MRI followed by ultrasound-guided punch biopsy was performed at this young age for further differential diagnostic clarification. Normally, this is not necessary.
Histopathological workup then revealed the typical findings of a benign vascular lesion with dysplastic, asymmetric irregular venous wall structure. The smooth muscular vessel wall is not formed throughout and is entirely missing in places. Histopathologically, another slow-flow vascular malformation, namely a lymphatic malformation, might be considered as a differential diagnosis. This could easily be differentiated with podoplanin stain (D2-40) to distinguish between venous malformation (D2-40-negative endothelia) and lymphatic malformation (D2-40-positive endothelia). Clinically, venous malformation was clearly more likely in this case because of the blue coloration of the lesion.
Directly initiated therapy took the form of just one session of bleomycin electrosclerotherapy (BEST). After only one session with intralesional injection of a minimal bleomycin dose (0.75 mg), there was a clear improvement of the venous malformation. There have been no complications, including no scarring.
Published: 2022
All images © Wohlgemuth/Dießel




![Venous malformation on the labium majus [Translate to English:] GenitaleVenous malformation on the labium majus venöse Malformation](/fileadmin/_processed_/6/f/csm_f43-15-venoese-malformation_cc3c0a8b76.jpg)
